

2025 , Vol. 22 >Issue 08: 761 - 767
DOI: https://doi.org/10.3877/cma.j.issn.1672-6448.2025.08.011
双表型肝细胞癌与经典型肝细胞癌超声造影特征及危险因素分析
通信作者:
卢强,Email:luqiang@scu.edu.cnCopy editor: 汪荣
收稿日期: 2025-03-29
网络出版日期: 2025-09-29
基金资助
山西省卫生健康委员会科研课题(2024118)
版权
Contrast-enhanced ultrasound features of dual-phenotype versus classical hepatocellular carcinoma and risk factors for dual-phenotype hepatocellular carcinoma
Corresponding author:
Lu Qiang, Email: luqiang@scu.edu.cnReceived date: 2025-03-29
Online published: 2025-09-29
Copyright
探讨双表型肝细胞癌(DPHCC)及经典型肝细胞癌(THCC)的临床病理特征及超声造影表现差异,并分析DPHCC的独立危险因素。
本研究是一项回顾性研究。纳入2019年1月至2024年6月经手术病理确诊的71例肝细胞癌患者,其中DPHCC 31例,THCC 40例。对比2组的临床病理特征及超声造影特征,采用多因素Logistic回归分析DPHCC的独立危险因素。
DPHCC组血清甲胎蛋白≥400 ng/ml的比例(48.4% vs 15.0%,P=0.002)及微血管浸润率(77.4% vs 17.5%,P<0.001)显著高于THCC组。超声造影显示,与THCC组相比,DPHCC组动脉期以不均匀增强(38.7% vs 15.0%)和环状增强(16.1% vs 2.5%)为主(P=0.003),且廓清时间更早[(91.94±37.58) s vs (131.65±59.71)s,P=0.002)]。多因素Logistic回归分析显示,肿瘤大小(OR=1.019,P=0.002)、甲胎蛋白≥400 ng/ml(OR=2.798,P=0.032)、边界不清(OR=3.204,P=0.020)及廓清时间<120 s(OR=2.221,P=0.049)是DPHCC的独立危险因素。
多参数超声造影成像可为临床无创诊断DPHCC提供重要依据,有助于鉴别DPHCC与THCC,优化个体化诊疗策略。
杨秀玲 , 王文辉 , 杨婕 , 卢强 . 双表型肝细胞癌与经典型肝细胞癌超声造影特征及危险因素分析[J]. 中华医学超声杂志(电子版), 2025 , 22(08) : 761 -767 . DOI: 10.3877/cma.j.issn.1672-6448.2025.08.011
To explore the differences in clinicopathological characteristics and contrast-enhanced ultrasound (CEUS) manifestations between dual-phenotype hepatocellular carcinoma (DPHCC) and typical hepatocellular carcinoma (THCC), and to identify independent risk factors for DPHCC.
This retrospective study enrolled 71 patients with pathologically confirmed hepatocellular carcinoma after surgical resection (31 cases of DPHCC and 40 cases of THCC) from January 2019 to June 2024. Clinicopathological parameters and CEUS features were compared between the two groups. Multivariate logistic regression was used to identify independent risk factors for DPHCC.
The DPHCC group showed significantly higher rates of serum alpha-fetoprotein (AFP) ≥400 ng/ml (48.4% vs 15.0%, P=0.002) and microvascular invasion (77.4% vs 17.5%, P<0.001) compared to THCC. On CEUS, DPHCC predominantly exhibited heterogeneous enhancement (38.7% vs 15.0%) and rim-like enhancement (16.1% vs 2.5%) in the arterial phase (P=0.003), with earlier contrast agent washout initiation (91.94±37.58 s vs 131.65±59.71 s, P=0.002). Multivariate analysis identified tumor size (odds ratio [OR]=1.019, P=0.002), AFP≥400 ng/ml (OR=2.798, P=0.032), ill-defined margin (OR=3.204, P=0.020), and washout time <120 s (OR=2.221, P=0.049) as independent risk factors for DPHCC.
Multi-parametric CEUS provides critical evidence for noninvasive diagnosis of DPHCC, effectively differentiating it from THCC and optimizing personalized clinical management.
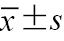
表1 DPHCC组与THCC组患者的一般临床资料及病理特征比较[例(%)] |
| 临床资料及病理特征 | DPHCC组(n=31) | THCC组(n=40) | 统计值 | P值 |
|---|---|---|---|---|
年龄(岁,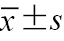 | 53.26±9.65 | 58.05±10.73 | t=1.98 | 0.055 |
| 女性 | 16(51.6) | 3(7.5) | χ2=17.32 | <0.001 |
病灶最大径(mm,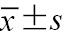 | 45.31±33.21 | 39.63±23.27 | t=0.81 | 0.396 |
| 肝炎病史 | 31(100) | 36(90.0) | χ2=1.31 | 0.252 |
| AFP(≥400 ng/ml) | 15(48.4) | 6(15.0) | χ2=9.35 | 0.002 |
| CA19-9(≥37 U/ml) | 5(16.1) | 7(17.5) | χ2=0.02 | 0.878 |
| PIVKA-Ⅱ(≥40 mAU/ml) | 21(67.7) | 27(67.5) | χ2=0.00 | 0.983 |
| CK7阳性率 | 14(45.2) | 0(0) | - | <0.001 |
| CK19阳性率 | 31(100) | 0(0) | - | <0.001 |
| 组织分化程度 | χ2=2.22 | 0.412 | ||
| 高分化 | 6(19.4) | 3(7.5) | ||
| 中分化 | 17(54.8) | 25(62.5) | ||
| 低及未分化 | 8(25.8) | 12(30.0) | ||
| MVI | 24(77.4) | 7(17.5) | χ2=25.51 | <0.001 |
注:DPHCC为双表型肝细胞癌;THCC为经典型肝细胞癌;AFP为甲胎蛋白;CA19-9为糖类抗原19-9;PIVKA-Ⅱ为异常凝血酶原;CK17为细胞角蛋白17;CK19为细胞角蛋白19;MVI为微血管侵犯;-表示Fisher检验无统计值 |
表2 DPHCC组与THCC组患者的灰阶超声及超声造影特征比较[例(%)] |
| 超声特征 | DPHCC组(n=31) | THCC组(n=40) | 统计值 | P值 |
|---|---|---|---|---|
| 内部回声 | χ2=0.25 | 0.883 | ||
| 强回声 | 6(19.4) | 9(22.5) | ||
| 弱回声 | 20(64.5) | 26(65.0) | ||
| 不均匀回声 | 5(16.1) | 5(12.5) | ||
| 边界 | χ2=0.52 | 0.471 | ||
| 清晰 | 20(64.5) | 29(72.5) | ||
| 不清晰 | 11(35.5) | 11(27.5) | ||
| 形态 | χ2=0.67 | 0.413 | ||
| 规则 | 18(58.1) | 27(67.5) | ||
| 不规则 | 13(41.9) | 13(32.5) | ||
增强时间(s,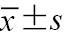 | 15.32±4.37 | 16.88±4.88 | t=1.14 | 0.169 |
廓清时间(s,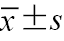 | 91.94±37.58 | 131.65±59.71 | t=3.41 | 0.002 |
| 动脉期增强方式 | - | 0.003 | ||
| 均匀增强 | 14(45.2) | 33(82.5) | ||
| 不均匀增强 | 12(38.7) | 6(15.0) | ||
| 环状增强 | 5(16.1) | 1(2.5) | ||
| 门脉期增强程度 | - | 0.060 | ||
| 高增强 | 1(3.2) | 4(10.0) | ||
| 等增强 | 4(12.9) | 13(32.5) | ||
| 低增强 | 26(83.9) | 23(57.5) | ||
| 实质期增强程度 | - | 0.157 | ||
| 高增强 | 1(3.2) | 0(0) | ||
| 等增强 | 0(0) | 3(7.5) | ||
| 低增强 | 30(96.8) | 37(92.5) | ||
| 内部灌注缺损 | χ2=0.13 | 0.722 | ||
| 无 | 27(87.1) | 36(90.0) | ||
| 有 | 4(12.9) | 4(10.0) |
注:DPHCC为双表型肝细胞癌;THCC为经典型肝细胞癌;-表示Fisher检验无统计值 |
表3 DPHCC的单因素Logistic回归分析结果 |
| 变量 | 参照组 | 比较组 | β值 | 标准误差 | Wald值 | P值 | OR值(95%CI) |
|---|---|---|---|---|---|---|---|
| 性别 | 女 | 男 | 1.409 | 1.229 | 1.313 | 0.252 | 4.092(0.374,45.522) |
| 年龄 | - | - | 0.103 | 0.060 | 3.012 | 0.083 | 0.901(0.801,1.011) |
| 病灶最大径 | - | - | 0.023 | 0.026 | 0.764 | 0.004 | 1.032(1.011,1.052) |
| AFP | <400 ng/ml | ≥400 ng/ml | 1.162 | 0.493 | 5.552 | 0.004 | 3.203(1.453,7.098) |
| 异常凝血酶 | <40 mAU/ml | ≥40 mAU/ml | 0.000 | 0.000 | 0.407 | 0.523 | 1.000(0.999,1.000) |
| CA-199 | <37 U/ml | ≥37 U/ml | 0.004 | 0.008 | 0.214 | 0.644 | 1.003(0.993,1.022) |
| 内部回声 | 均匀 | 不均匀 | 1.226 | 0.502 | 5.968 | 0.015 | 2.496(1.202,5.199) |
| 边界 | 清晰 | 不清晰 | 1.335 | 0.432 | 9.541 | 0.002 | 3.802(1.603,8.998) |
| 动脉期强化 | 均匀 | 不均匀 | 0.741 | 0.352 | 4.432 | 0.036 | 2.098(1.052,4.200) |
| 门脉期强化 | 非低增强 | 低增强 | -1.059 | 1.302 | 0.661 | 0.416 | 0.352(0.033,4.445) |
| 实质期强化 | 非低增强 | 低增强 | 23.455 | 15 880.980 | 0.000 | 0.999 | - |
| 廓清时间 | ≥120 s | <120 s | 0.812 | 0.315 | 6.641 | 0.010 | 2.253(0.204,0.799) |
| MVI | 阴性 | 阳性 | 4.546 | 1.481 | 9.423 | 0.002 | 94.233(5.168,1716.701) |
注:DPHCC为双表型肝细胞癌;AFP为甲胎蛋白;CA19-9为糖类抗原19-9;MVI为微血管侵犯;-为连续性变量未赋值或无相关数据 |
表4 DPHCC的多因素Logistic回归分析结果 |
| 变量 | 参照组 | 比较组 | β值 | 标准误差 | Wald值 | P值 | OR值(95%CI) |
|---|---|---|---|---|---|---|---|
| 年龄 | - | - | 0.103 | 0.060 | 3.012 | 0.083 | 0.899(0.801,1.011) |
| 病灶最大径 | - | - | 0.019 | 0.006 | 9.210 | 0.002 | 1.019(1.007,1.032) |
| AFP | <400 ng/ml | ≥400 ng/ml | -1.704 | 0.687 | 6.148 | 0.032 | 2.798(1.103,7.147) |
| 内部回声 | 非低回声 | 低回声 | 0.741 | 0.432 | 2.941 | 0.087 | 2.099(0.901,4.898) |
| 边界 | 清晰 | 不清晰 | 1.964 | 1.512 | 1.688 | 0.020 | 3.204(1.195,8.503) |
| 动脉期强化 | 均匀 | 不均匀 | 0.585 | 0.410 | 2.038 | 0.150 | 1.796(0.799,3.998) |
| 廓清时间 | ≥120 s | <120 s | 0.798 | 0.406 | 3.863 | 0.049 | 2.221(1.002,4.923) |
| MVI | 阴性 | 阳性 | 3.968 | 1.042 | 14.503 | <0.001 | 0.019(0.002,0.146) |
注:DPHCC为双表型肝细胞癌;AFP为甲胎蛋白;MVI为微血管侵犯;-为连续性变量未赋值 |
| 1 |
中国抗癌协会肝癌专业委员会, 中华医学会肝病学分会肝癌学组, 中国抗癌协会病理专业委员会, 等. 原发性肝癌规范化病理诊断指南(2015年版) [J]. 中华肝胆外科杂志, 2015, 21(3): 145-151.
|
| 2 |
王瀚, 丛文铭. 双表型肝细胞癌新亚型的临床病理学研究进展 [J]. 中国肿瘤临床, 2017, 44(12): 616-619.
|
| 3 |
|
| 4 |
|
| 5 |
|
| 6 |
马妍, 王玉红, 林燕蝶, 等. 双表型肝细胞癌的临床病理特征分析及其与临床预后的关系 [J]. 诊断病理学杂志, 2022, 29(9): 806-809.
|
| 7 |
郑巧灵, 冯昌银, 连渊娥, 等. 双表型肝细胞癌六例临床病理学分析 [J]. 中华病理学杂志, 2020, 49(12): 1320-1322.
|
| 8 |
史连国, 方庆全, 陈宏. 双表型肝细胞癌的临床病理特征 [J]. 中国实用医刊, 2021, 48(10): 13-16.
|
| 9 |
|
| 10 |
王玺. 基于生物信息学探讨肝细胞癌CK19相关分子机制的研究 [D]. 南宁: 广西医科大学, 2021.
|
| 11 |
|
| 12 |
|
| 13 |
冉琪, 胡亚飞, 游玉峰. 超声造影表现与肝硬化背景下肝细胞癌组织病理、分化程度及预后的关系分析 [J]. 安徽医药, 2021, 25(4): 681-685.
|
| 14 |
|
| 15 |
|
| 16 |
|
| 17 |
张杰. 双表型肝细胞癌的诊断方法以及双表型与早期复发的相关性研究[D]. 南宁: 广西医科大学, 2018.
|
| 18 |
延永琴, 曲利娟, 叶显宗, 等. 细胞角蛋白19表达与双表型肝细胞癌临床病理特征及其与预后的相关性 [J]. 中华肝脏病杂志, 2025, 33(1): 48-56.
|
/
| 〈 |
|
〉 |